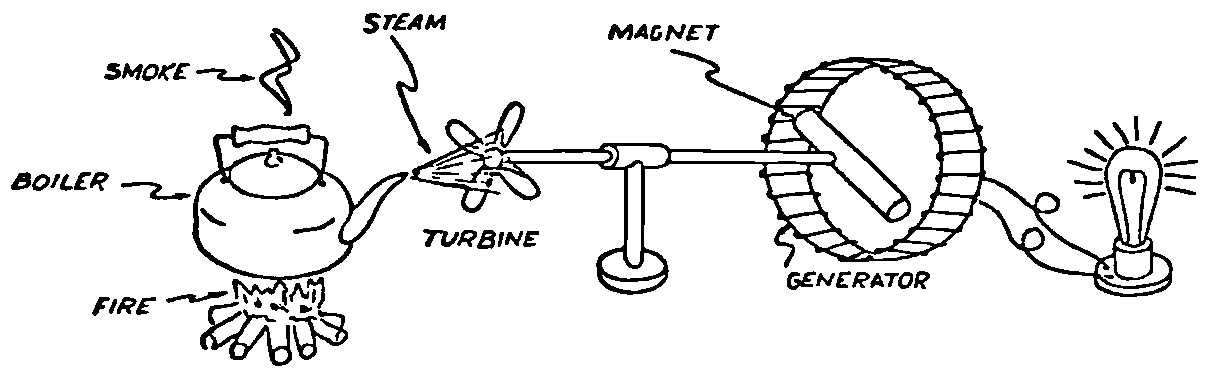
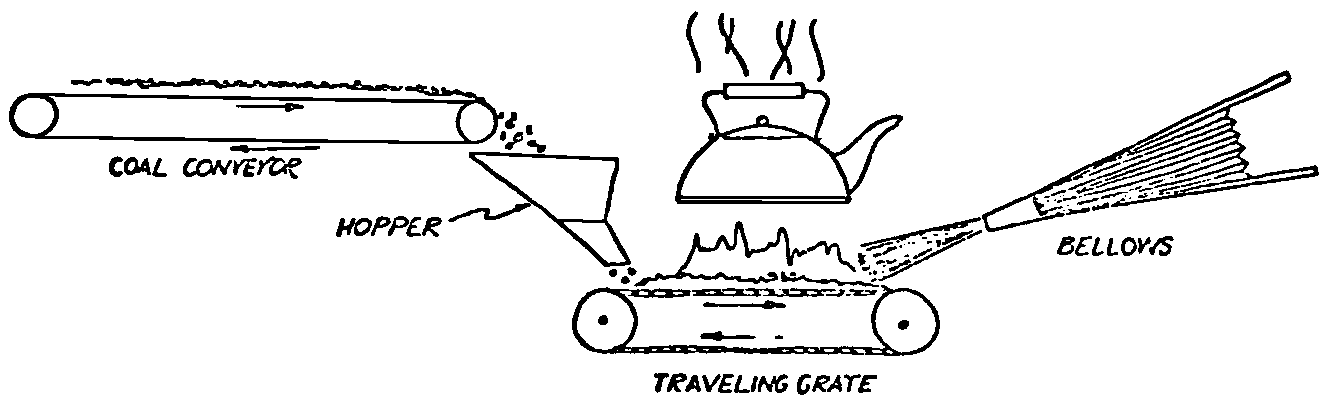
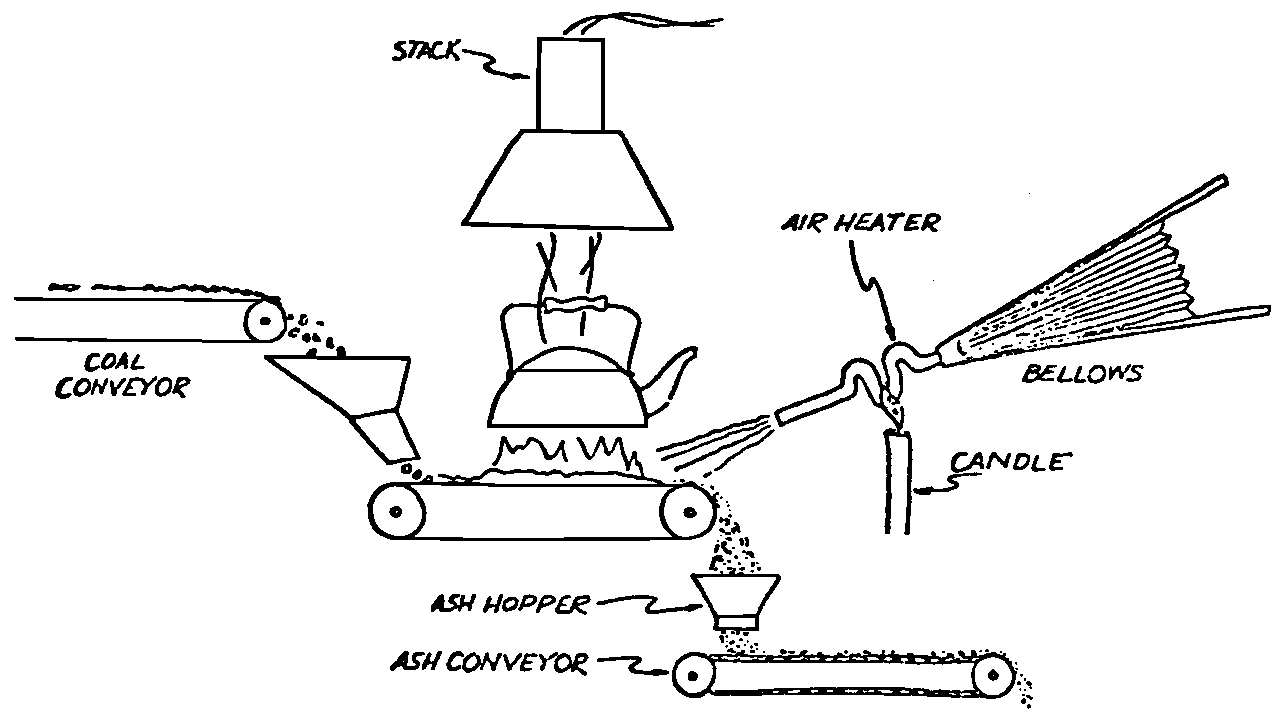
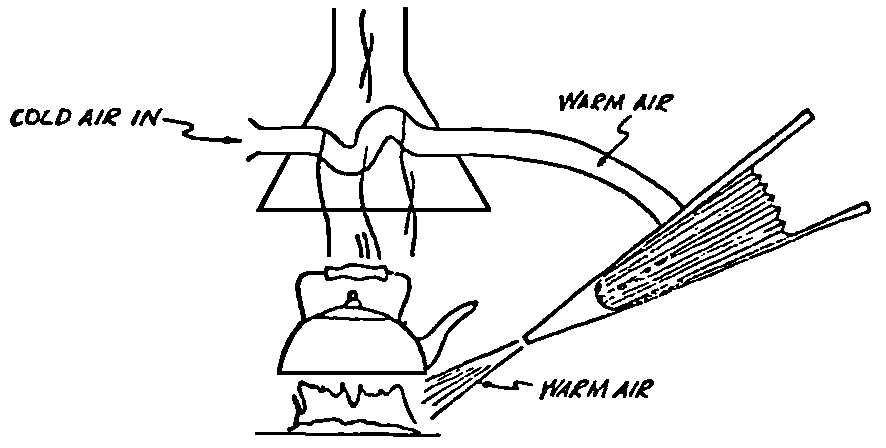
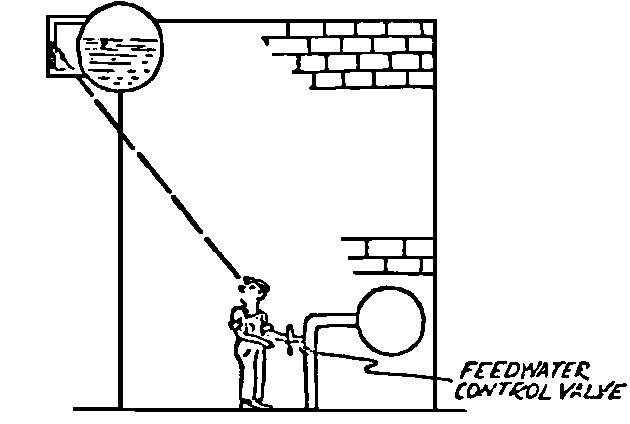
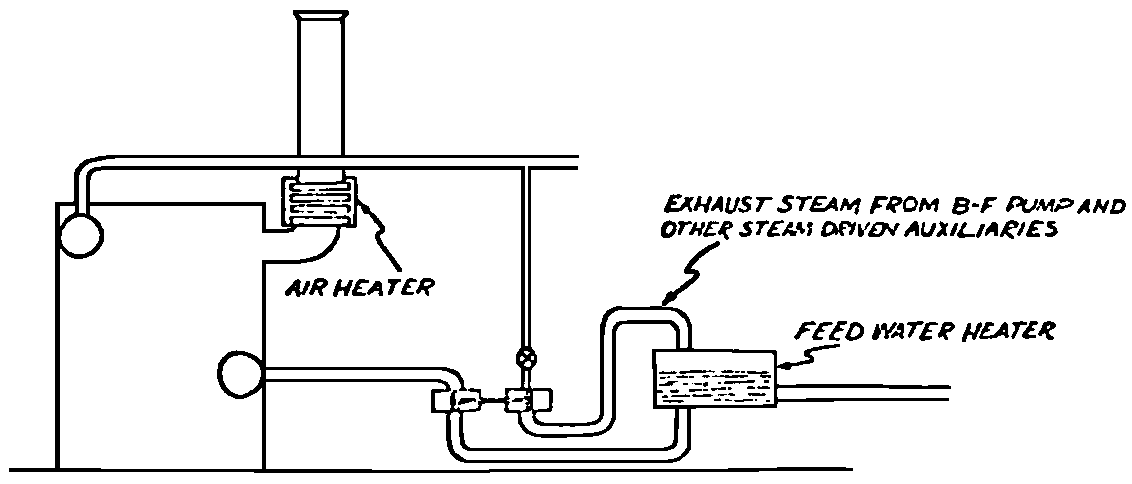
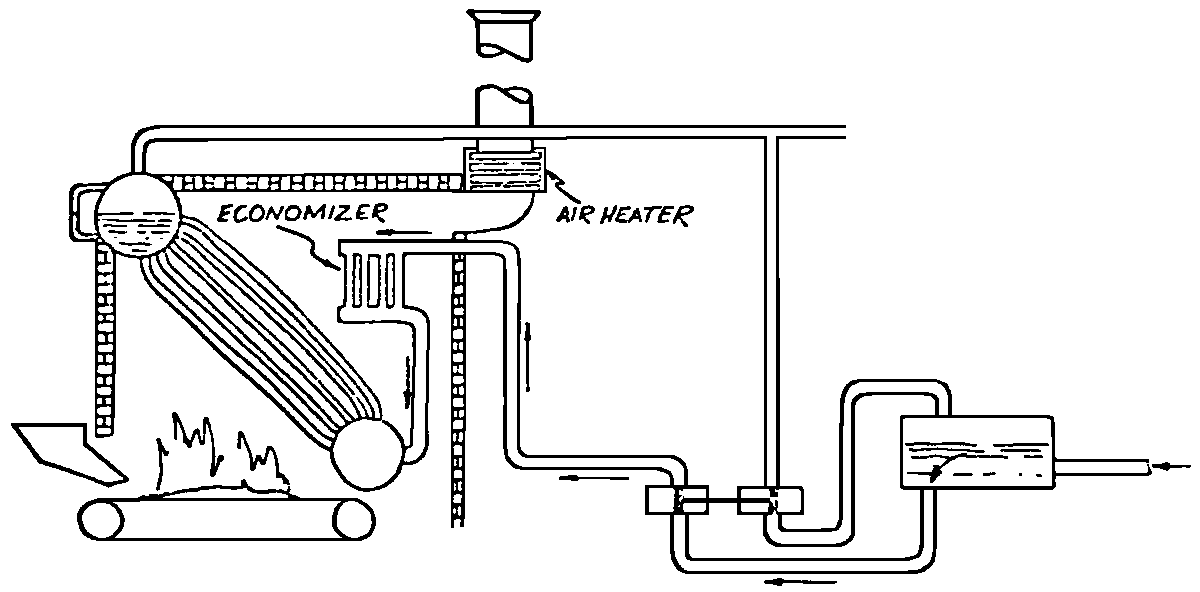
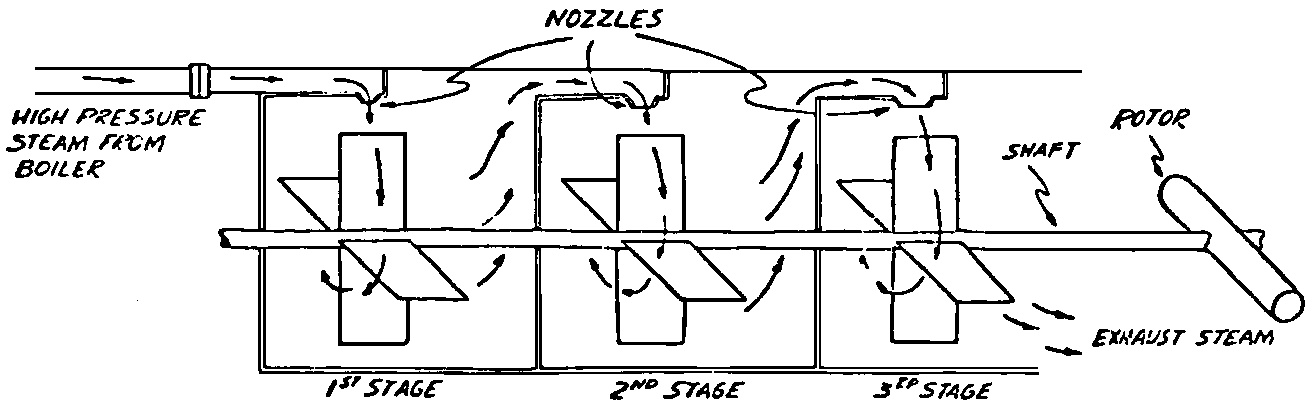
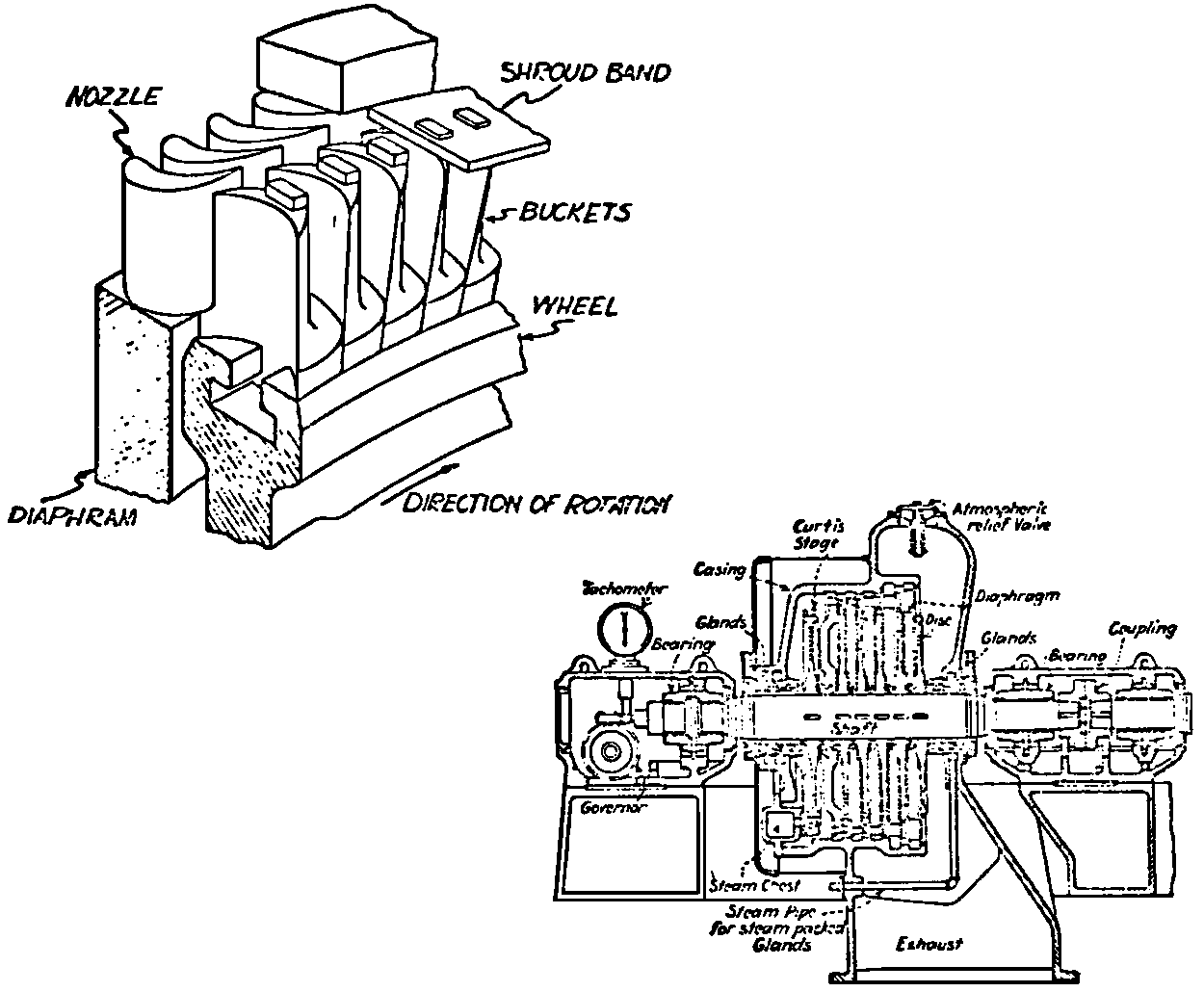
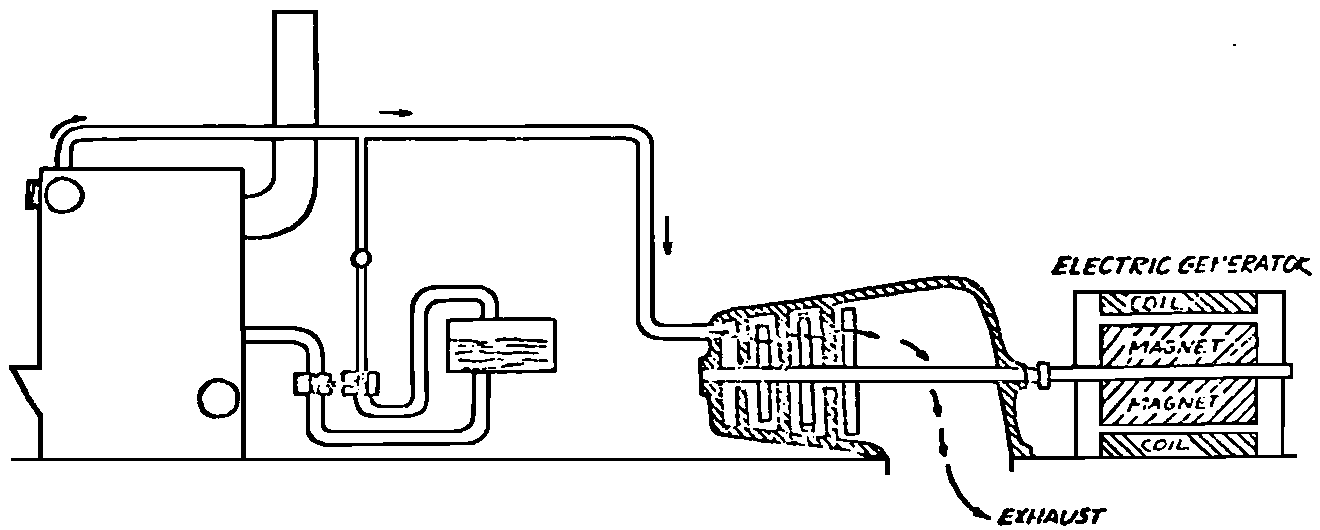
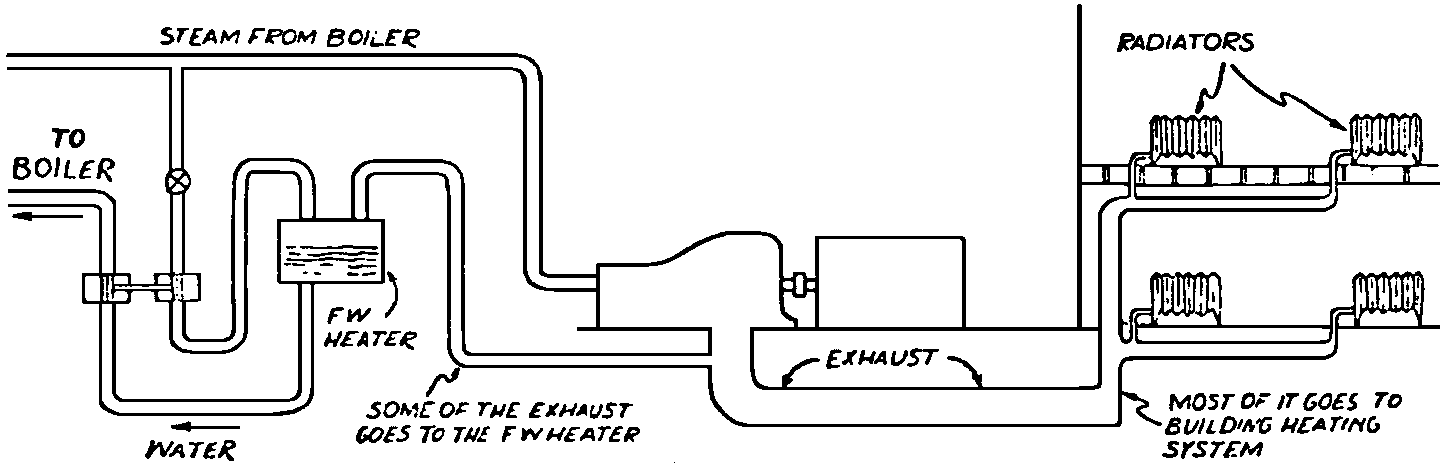
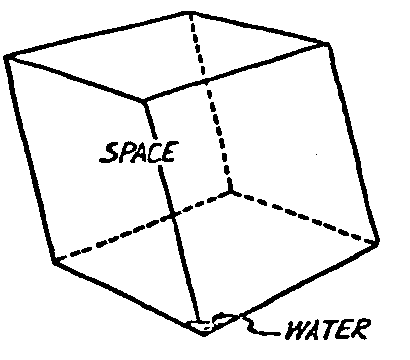
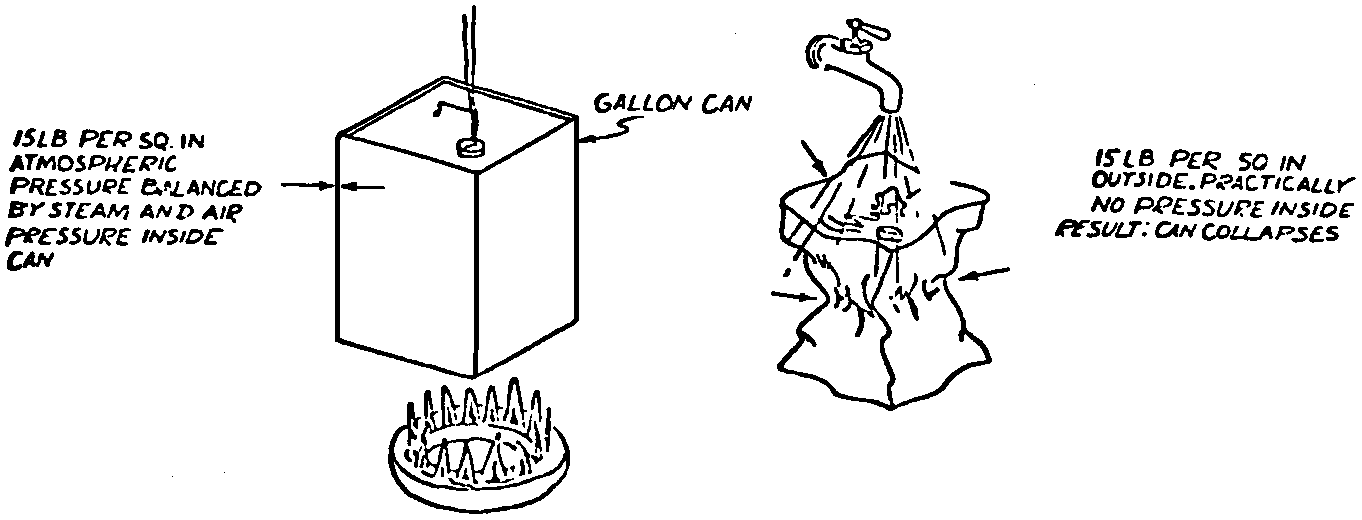
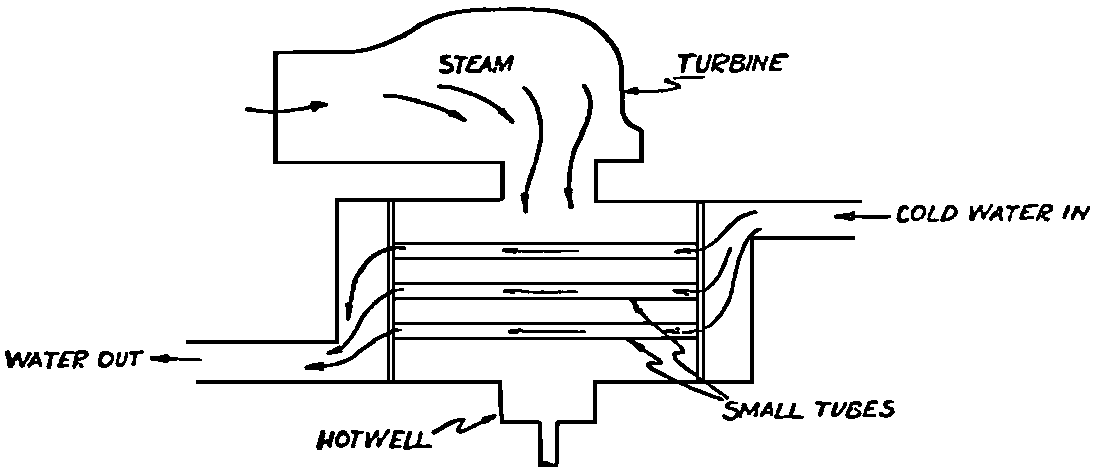
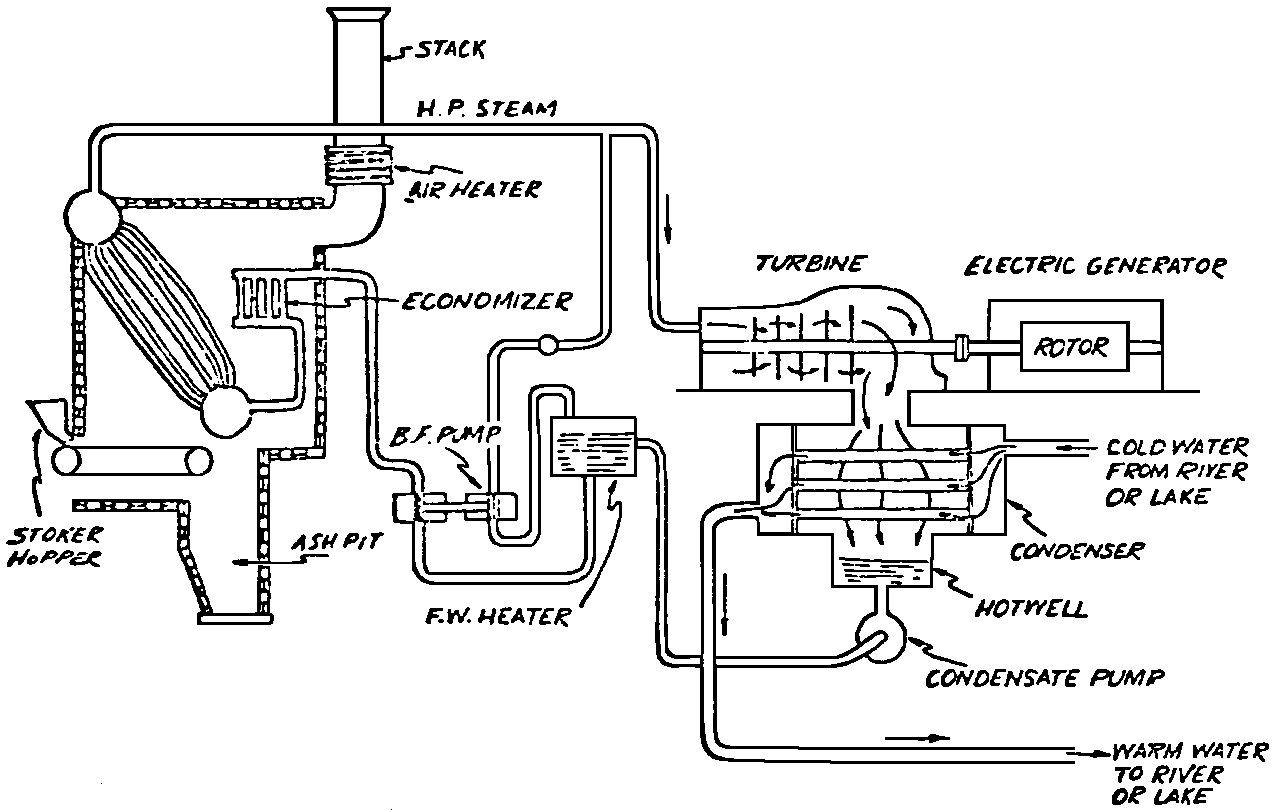
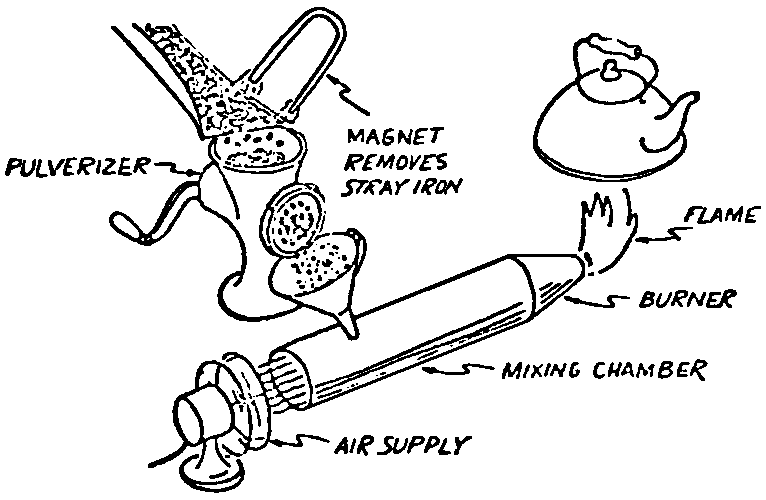
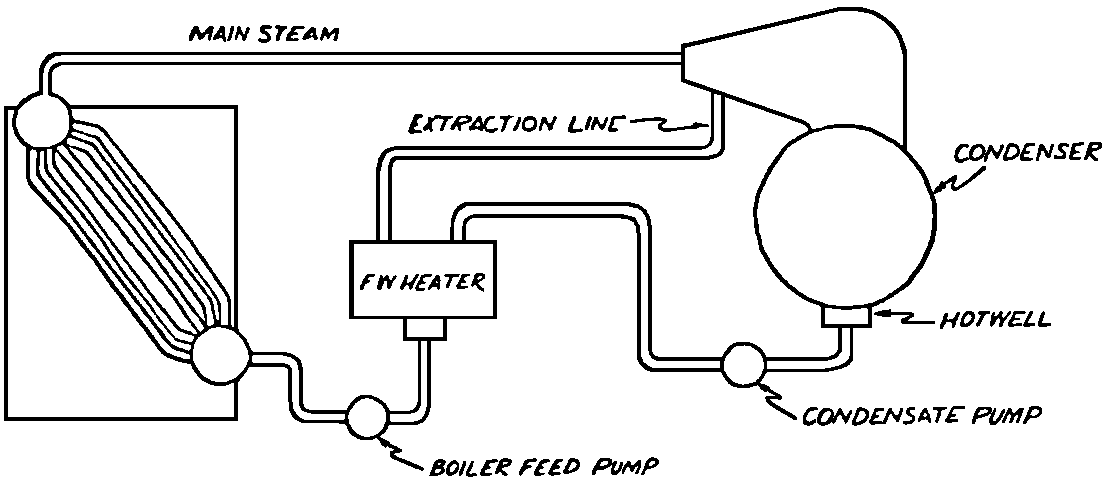
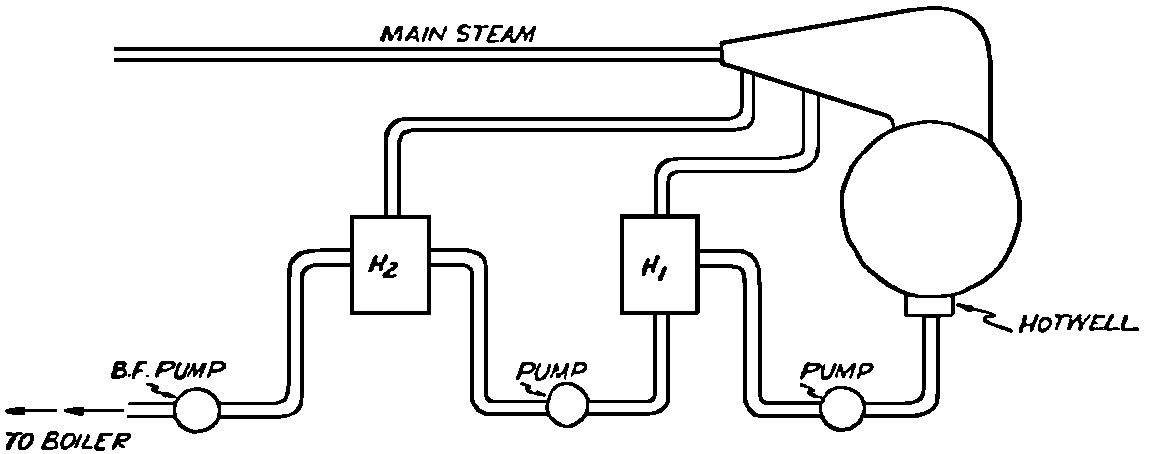
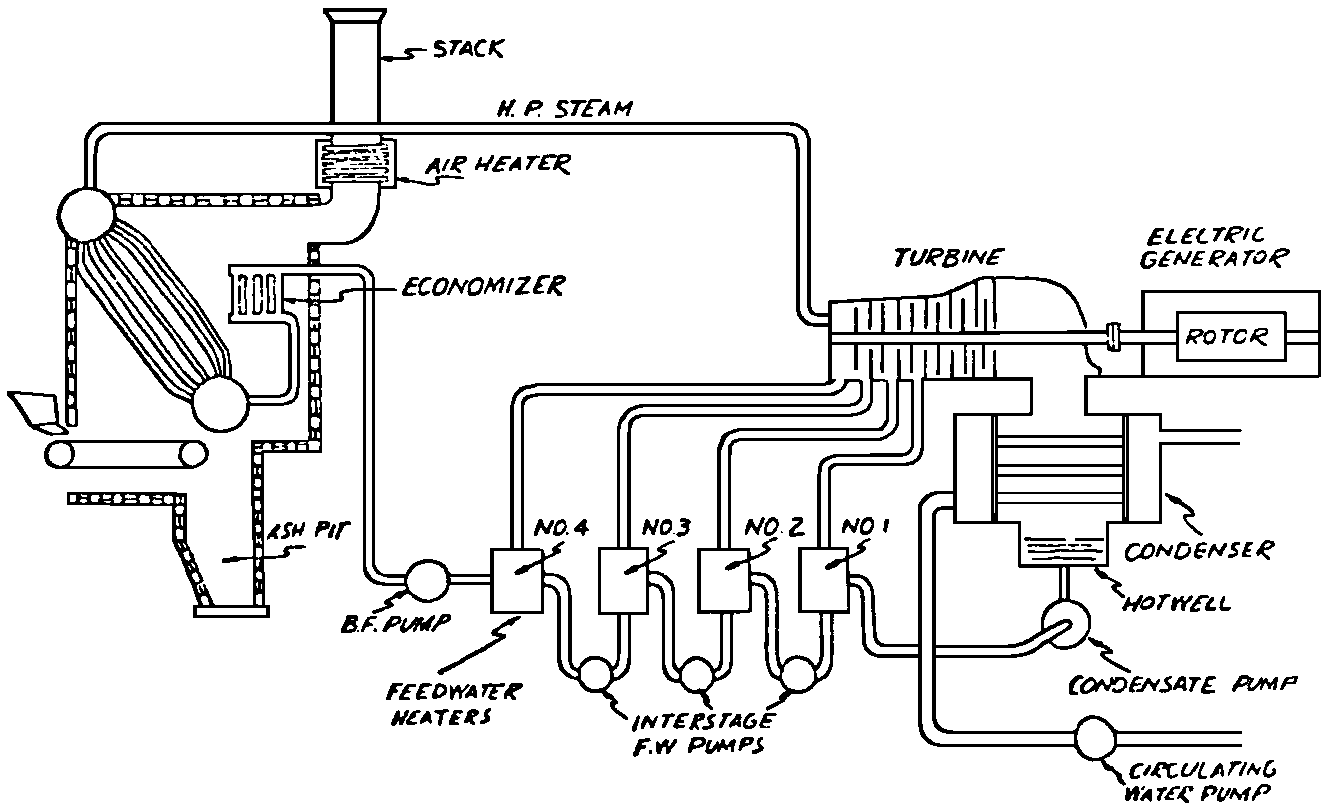
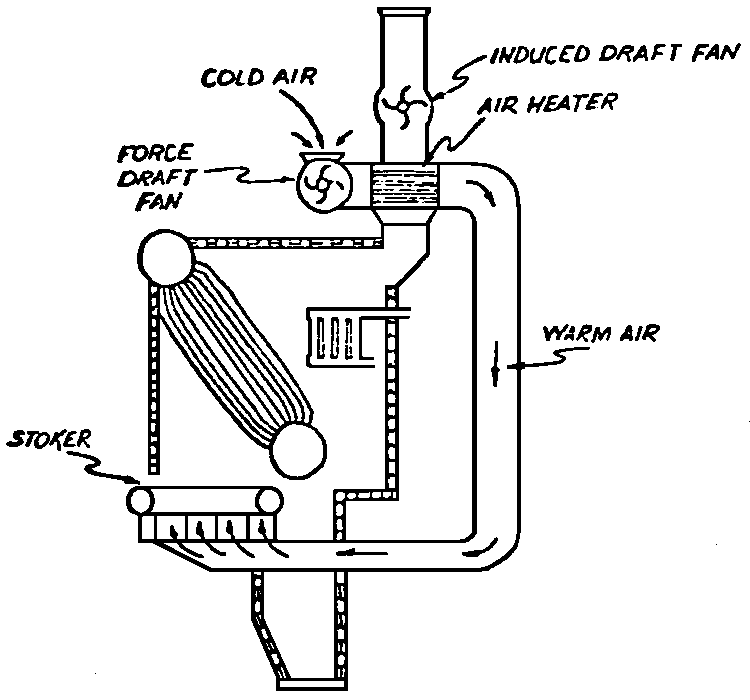
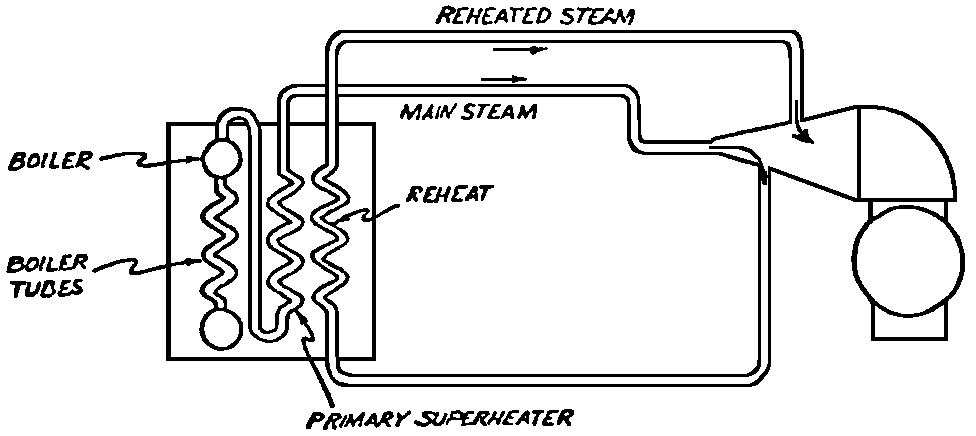
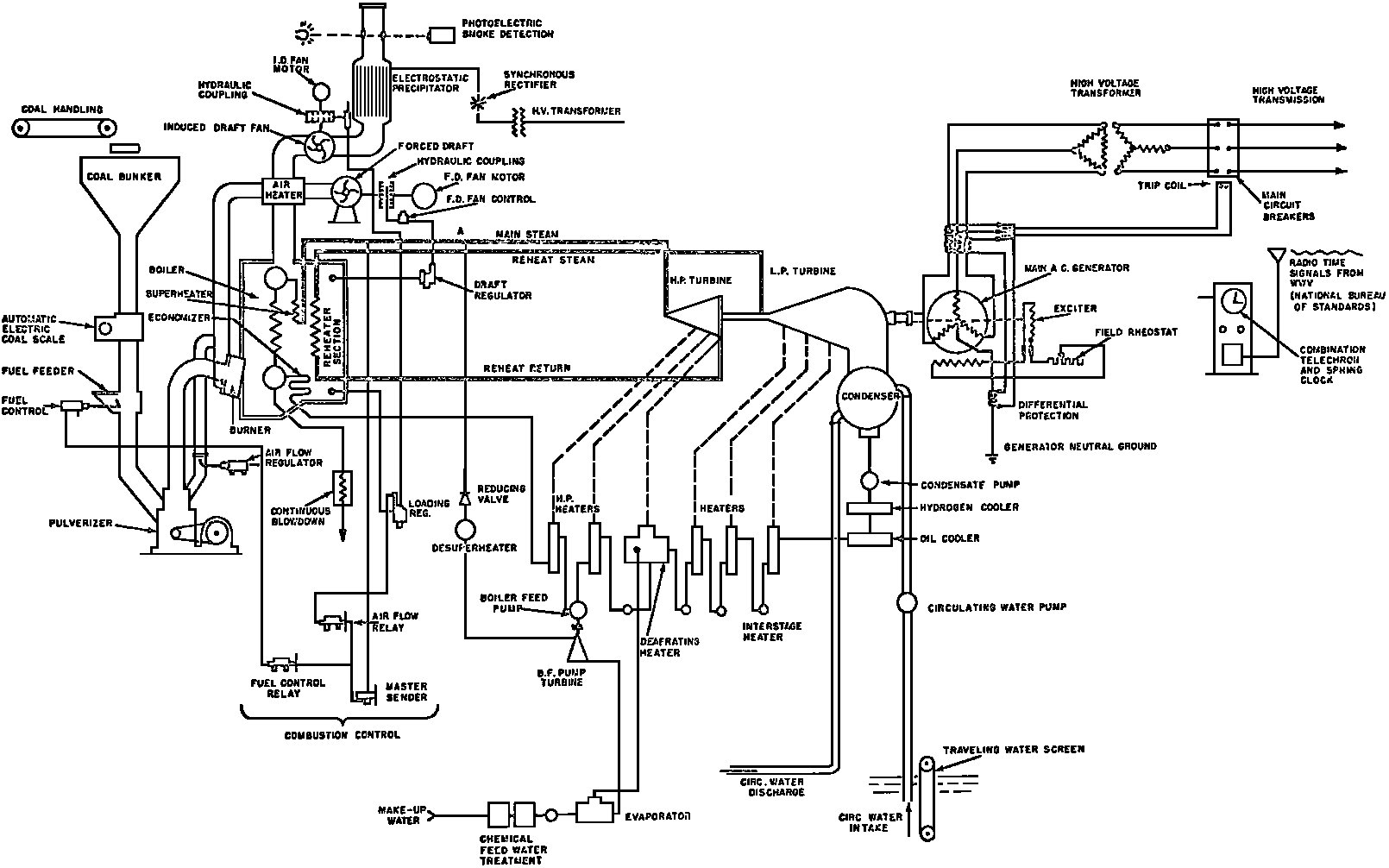
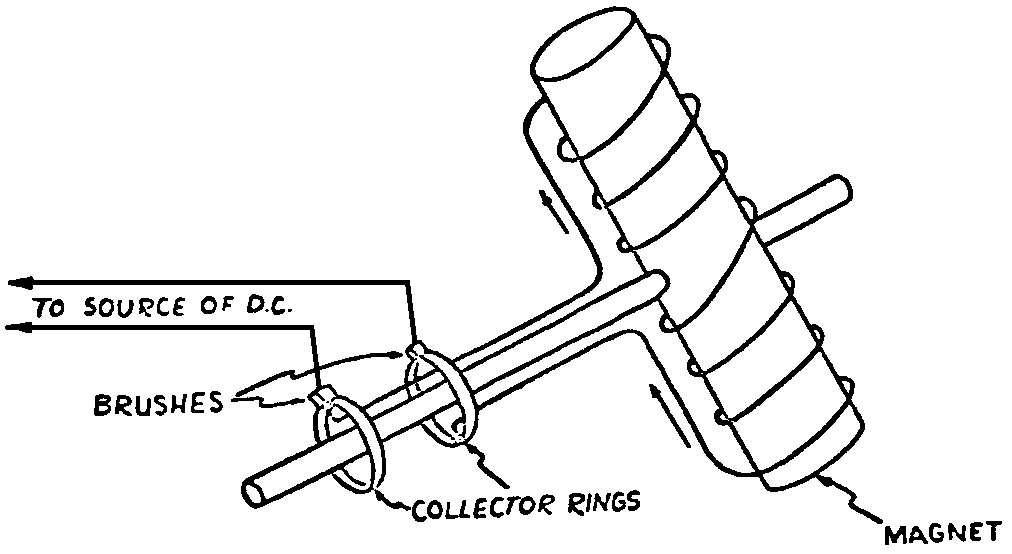
A Power Plant Primer A steam power plant is a means for converting the potential chemical energy of fuel into electrical energy. In its simplest form it consists of a boiler and a turbine driving an electric generator. The boiler is a device for turning water into steam. The steam jet issuing from the spout spins the fan (turbine) and also the generator. In the sketch the very simplest kind of boiler and turbine are shown. The boiler is a teakettle, and the turbine is nothing more than a little windmill. Actual turbines are more complicated but the principle is the same. A word about the generator shown in the sketch. To most people the process of generating electricity is very mysterious, yet the actual process is easy to understand. As shown, the generator consists of a little bar magnet spinning inside a stationary coil of wire. This may seem an absurdly simple affair, yet that is exactly what a real generator consists of – a magnet rotating inside of a coil of wire. As the magnet field issuing from the ends of the magnet mover across the turns of wire in the stationary coil an electric current is set up in the wire. By winding a large number of turns of wire into a ring or doughnut, the current set up in each turn is added to the current set up in the other turns of wire, and so a much more powerful current is produced. This is all you need to know about an electric generator now – just think of it as a rapidly rotating magnet inside of a coil of wire; this produces an electric current in the wire. Later we will elaborate on this simple description. You may wonder why, if a power plant is basically as simple as this, why do we build the complex plants we see described in POWER ENGINEERING? The answer to this question is quite simple; the plant shown in the sketch is not very efficient, indeed its efficiency is close to zero, and since we want to get as much power as possible out of a given quantity of fuel it is necessary to make our plants as efficient as we possibly can. Thirty years ago, the electric power plants of the nation used over 3 pounds of good coal to produce a kilowatt-hour of electricity. Today, the national average is less than 1 lb. of coal per kilowatt-hour. In other words, in 1922 we used three times as much coal to produce a kilowatt-hour as we use today. Last year, in 1955, the electric utilities of this country produced over 544 billion kilowatt-hours by means of fuel-fired plants. This required the burning of 272,000,000 tons of coal. If, however, we had had to produce this amount of electrical energy by means of the type of plants we had in 1922, we would have needed three times the coal or some 816,000,000 tons. The reason for this great decrease in the consumption of coal lies in the gradual improvement of our power systems, both with respect to the individual pieces of equipment and in the system as a whole. Just, how do we go about improving the system shown in the sketch? Looking at it again, it is obvious that it can be broken down into several divisions. First, there is the fire under the boiler. This involves not only the fuel itself but also the method of placing the fuel under the boiler and the arrangement for burning it properly. So let us extend the diagram to look like this: Here, we see a belt conveyor transporting coal to the furnace where it is burned on a traveling grate stoker. Air for combustion is supplied by a bellows. Remember, when you burn coal you are really promoting a chemical reaction – a chain reaction. When coal is heated to a high enough temperature in the presence of air, the carbon in the coal combines with oxygen of the air to form either carbon dioxide (CO2) or carbon monoxide (CO). These, of course, are both gases. Which gas is formed depends upon the quantity of oxygen present. The CO means that the coal is only partially burned; the CO can be combined with more oxygen to form CO2. In burning coal we do not want CO because that means the coal is only partially burned: there is still energy left in the gas, energy that we can recover if we can burn it to CO2. It is desirable, in the operation of our boiler furnaces, always to get as much CO2 as we can because in that way we get all the heat out of the fuel. Now we can obtain the CO2 by supplying more air to the fuel as it is burning. But, we do not want to supply too much air because if we do, we will be supplying more oxygen than is actually needed to combine with carbon, and this excess oxygen will play no part in the combustion process. Not only will it play no part but it will actually detract from the efficiency by absorbing heat that otherwise could be used to heat the water in the boiler. In actual practice it is not possible to supply exactly the required amount of air, so somewhat more than enough is supplied. This is commonly referred to, as excess air. So, in the process of combustion, we are dealing with chemistry. Thus, it involves knowledge of the composition of the coal, its physical condition, its behavior under various conditions of temperature, moisture, etc. Actually, the combustion of coal is a very complex process requiring a good knowledge of both physics and chemistry. In a large plant it involves a major problem in materials handling – fuel, ashes, air and flue gas. Remember, in burning coal, you have to supply about 17 pounds of air for each pound of coal used. So far, we have merely mentioned ashes and flue gas. These have to be removed continuously. In the days of hand firing the removal of ash was simple though laborious. The fireman merely raked the ashes out of the ash pit and carried it away in wheelbarrows. Today, in large plants, the removal of ash is a complicated process requiring rather elaborate equipment. So, we must add ash removal equipment to our diagram; also a chimney for the removal of flue gas. Furthermore, the process of combustion is stimulated by heat; indeed, the process will not start until the fuel is brought to the kindling temperature. Everything must be done, therefore, to maintain a high temperature in the furnace. This makes it desirable to heat the air for combustion before it is delivered to the furnace. This aids combustion and increases the efficiency. With these facts in mind, let us redraw our diagram to incorporate these improvements. This way: As you see, it is becoming more complicated. Now, we have a chimney or stack to remove the gas of combustion and a heater to heat the air from the bellows before it is blown into the furnace. Also, we have put in an ash conveyor. Now, if you are of an analytical mind, you will see that it takes additional heat to heat the air. We have shown a candle. Since candles cost money, it is obvious that we are not going to save much money that way. Why can't we use some of the heat from the fire under the boiler itself to heat the air? Maybe that would be cheaper than buying candles? Well, it is, and moreover, there is heat going to waste up the stack. You know from experience that if you hold your hand above a teakettle on a stove that there is a lot of heat being wasted. So, let us put a heating coil in the stack so the bellows will be blowing warm air into the fire as shown in the following sketch. All this, even to non-engineers, probably seems absurdly simple, and it is. The reason for explaining it in this way, however, is to show, by means of the simplest kind of equipment, how engineers go about improving the efficiency of any system. Step by step, adding something here, saving something there, establishing closer supervision over everything gradually improves the effectiveness and the efficiency of almost any kind of system. These are the kind of things engineers are concerned with. Nearly all of them are more or less complex and require a great deal of specific as well as general knowledge. In the example just described, for example, just how much surface should the heater in the stack have to heat the air to a certain temperature? How hot should the air be for best combustion, how much air should be supplied, how much power will it take to run the bellows, what happens to the flue gases if too much heat is extracted from the gases, what happens to the stack? None of these is a foolish question. Consider the last question, for example, that of cooling the flue gases too much. What happens? Well, there is always a certain amount of water vapor in the flue gases, from the air and from the hydrogen in the fuel. As the temperature of the gases is lowered, there comes a time when the saturation point is reached and the moisture condenses. If, at the same time there happens to be any sulfur in the gas, (and there usually is) sulfuric acid will be formed resulting in the spread of a thin but extremely corrosive layer of liquid on the inside surfaces of the flues. This single example, then, which is one among hundreds, shows what the engineer runs into when he begins to refine the simple system we began with. He may add something to improve it but he may find that the improvement is not an unmixed blessing; it may also have deleterious effects. These, he must guard against. But, let's get on with our power system. The teakettle representing the boiler is not a very efficient generator of steam. Let's see if we cannot design a better one. Look at the following sketch. Here we have a boiler consisting of two steel drums connected by a number of steel tubes, and arranged in a furnace so that the hot gases have to pass through the bank of tubes on their way to the stack. The total surface of the tubes is large, making it possible to absorb a great deal of heat. The steam bubbles formed in the tubes rise to the upper drum (called the steam drum) where the steam collects before it flows into the pipe leading to the turbine. This is the basic principle of the modern water tube boiler. Note, that a boiler feed pump has been included in the diagram. Since steam flows out of the boiler it obviously becomes necessary to replenish the water that is evaporated. For this reason a boiler feed pump is necessary. This pump must operate at a pressure high enough so that it can overcome the pressure in the boiler. In the operation of any boiler, even a teakettle, it is essential always to keep enough water in the boiler. If it should run dry the metal would become red hot, soften, and rupture. At the same time it should not be filled to a point where there is no room for the steam to collect. To check the water level, a water gage is fitted to the steam drum. This, at all times shows the water level in the boiler. Now, it is obvious that if the amount of steam leaving the boiler was always the same as the amount of water entering, the water level would remain the same. This state of affairs seldom transpires. Variations in load cause variations in steam flow; variations in the fuel supply and air supply cause variations in the rate of combustion, which in turn results in variations in the rate of evaporation, and all of these cause change s in the water level. This makes it necessary for the operator to maintain a continual watch on the water level. If it drops, he increases the water supply; if it rises, he decreases it. This constant vigilance on the part of operators was trying, so feedwater regulators were developed which control the flow automatically as the water in the boiler drum rises and falls. These are very helpful even with small boilers but in the case of modern high-pressure boilers they are almost imperative. A large high-pressure boiler, evaporating in the neighborhood of a million pounds of water per hour would run dry in about 90 seconds if the water supply was suddenly cut off. Now, getting back to our water supply to the boiler, so far nothing has been said about the temperature of the water being delivered to the boiler. It should be obvious, however, that it would not be wise to pump cold water into a boiler since that would decrease the temperature of the water already in the boiler and so reduce the rate at which steam was being made. Also, the introduction of cold water might set up strains in the boiler by virtue of the great temperature difference. It, therefore, becomes expedient to heat the water to as high a temperature as possible before pumping it into the boiler. So, it is run through a feedwater heater. The feedwater heater could be heated by a separate fuel-fired furnace but, as in the case of the air beater, it would be much more economical if it could be heated by heat that would otherwise be wasted. Suppose, for example that the boiler feed pump was a steam driven pump, and we could use the exhaust steam from this pump. This would cost virtually nothing. So, we add a heater as shown here. After the steam has done its work driving the pump, it is delivered to the feedwater heater, which, as shown here, is nothing more than a large tank open to the atmosphere. Hence, it is called an open heater. As will be shown later, there are other kinds of feedwater heaters called closed heaters. These usually operate at pressures above atmospheric pressure. So far, so good, we have saved a little by using the heat in exhaust steam which otherwise would have been wasted. Let's look a bit further however; maybe we can save some more heat somewhere else. Remember, we picked up some heat in the flue gases by means of the air heater. Have we got all of it? Whether we have or haven't is not so much of importance, because, if we want we can modify the boiler so as to set aside one section of the tubes to heat the water before it is delivered to the boiler proper. Look at this sketch: Here we have added a separate bank of tubes through which the feed water passes before it goes into the boiler drum. This bank of tubes is placed in the path of the gases traveling, towards the air heater and the stack. Most of the heat in these gasses has been absorbed in the boiler tubes but not all of it. They probably still have a temperature of around 600 degrees. By making them travel through this added tube bank still more of the heat will be absorbed, consequently the economy of the boiler as a whole will be increased. Hence, this bank of tubes is known as an economizer. With this arrangement, the water is first heated to a temperature of around 212 degrees in the feedwater heater by the exhaust steam from the feed pump, and then, in the economizer, the temperature is further raised to a point not very far below the temperature of the water in the boiler. Our boiler, or steam generator as it is called these days, has now become quite complex so before we do anything more to it let's hook it up to the rest of the system. As already mentioned a turbine is essentially a windmill, not a simple fan like the one shown but a more complex one with many hundreds of blades, some stationary, and some rotating. These blades are arranged in groups or stages, so that the steam is compelled to pass successively through the various stages. Here is a very simple diagram. In this diagram, three fans are shown mounted on a common shaft, each one in a separate compartment. Steam issuing from the nozzle in the first stage pushes against the fan blades and causes the entire rotor assembly to turn. In turning the blades in the first stage the steam gives up some of its energy, resulting in a drop in pressure. Thus, at a slightly lower pressure, it enters the second stage nozzle, and again it gives up some of its energy in turning the second stage of this rotor. After passing through the third stage in this way, practically all of the energy of the steam has been given up to the rotor and it leaves the turbine as exhaust steam. This arrangement, it should be obvious, provides a much more efficient means for spinning the rotor shaft than the simple little fan shown in the first diagram. Of course, it is still merely a diagram – no turbine would ever be built that way. The only step remaining to make this elemental turbine into a commercial machine is to introduce multiple nozzles of proper design and change the shape of the inefficient paddles to an efficient blade having curved entrances and exits. Here is a detailed drawing of the nozzles and blades (they are also called buckets) of a modern turbine, and next to it is a cross-section through a turbine showing how the elements are arranged on the shaft. So, now we have a boiler and a turbine coupled to an electric generator in this fashion. Here you see steam from the boiler being fed into the turbine by means of the connecting piping and after passing through the various stages of the turbine the steam exhausts through an opening in the bottom of the turbine. The steam has given up its energy to the turbine rotor and this in turn spins the generator rotor. The generator rotor, remember, is simply a magnet. But what shall we do with the exhaust steam? Is it of any use? Well, if you measure the temperature of the exhaust steam right at the point of the exhaust opening, you will find that it has a temperature of 212°F. This is the temperature of steam at atmospheric pressure. Obviously, we can use it to heat the water in the feedwater heater in the same way that we used the exhaust steam from the boiler feed pump. However, you would find that there is far more exhaust steam coming from the turbine than you could use in the feedwater heater. Remember, practically the entire boiler output passes through the turbine and out into the exhaust. Well, if you want you can pipe this exhaust steam to radiators and use it to heat our houses and buildings in winter, this way: Note that part of the exhaust steam goes to the feedwater heater. Most of it, however, goes to the building heating system. Now, this is fine if you have a building you want to heat or if you need steam heat for other purposes such as cooking, heating stills in food or chemical plants or any of scores of different purposes in industry, and in practice that is how a great deal of exhaust steam is used. Indeed, this is one of the reasons it pays the owners of an industrial plant, a paper mill for example, or a textile mill or a food products plant, to have their own power plant; they can use practically all of the exhaust steam from the turbine for heating purposes. If they did not have the turbine, they still would need a boiler to generate steam. By first running the steam through a turbine they can get the electric power so produced virtually for nothing. Suppose however, there is no building to be heated or no factory process to use up the exhaust steam, what then? Take a public utility plant for example. A public utility plant is designed solely to generate electricity to sell. Such plants usually are far away from buildings where the exhaust steam might be used for heating. Shall the exhaust steam be permitted to be wasted to the atmosphere? No. There is a much better way of disposing of this exhaust steam. That is by using it to create a vacuum at the exhaust end of the turbine. A vacuum? Why a vacuum? What good would that do? Well, remember that the turbine is surrounded on all sides by the atmosphere, which, at sea level, exerts a pressure of about 15 lb. per square inch. In order to get out of the exhaust opening, the steam has to push against this 15 lb. per sq. in. pressure, and this requires work, just as it does to push the turbine blades around. But suppose, by some means we could remove the atmosphere from around the exhaust opening so that the steam issuing from the exhaust opening would encounter no resistance whatever; you would find that you could develop more power in the turbine, indeed, it would be equivalent to an increase in steam pressure. To understand how we can accomplish this (removal of the atmospheric pressure) it is necessary to know a few facts about steam. Steam, remember, is evaporated water. When water is heated to a temperature of 212°F at atmospheric pressure it turns into steam. If the water is enclosed in a tightly closed vessel such as a boiler the temperature at which the water turns into steam will be higher. In any case, the volume of the steam produced will be very much larger than the volume of the water from which it was produced. At atmospheric pressure, for example, a pound of steam occupies a volume of 26 cubic ft. Suppose, now, that you had a pound of steam at atmospheric pressure in a closed vessel with a volume of exactly 26 cu. ft. This vessel would be a trifle less than 3 feet on a side – assuming it to be a cube. It would be full of steam. There would be no air. If you suddenly placed this vessel on a large block of ice, or cooled it by spraying cold water on it, what would happen? The steam would condense – it would turn back into water – into one pound of water. This pound of water, however, would occupy only 1/60th of a cubic foot. It would look about like this: This is very little water. Most of the interior is now occupied by nothing – 99.93 percent of the total volume. This means a vacuum. The total surface of this cube bas an area of 7,776 sq. in. Since each square inch bas 15 lb. of atmosphere pressing down on it (and with nothing inside to counteract it) the total atmospheric pressure on the cube is now 7,776 × 15 or about 116,640 lb. If you want to see whether this is really true, try it sometime. Take an ordinary rectangular gallon can with a screw cap closure, fill it with about half inch of water, and bring the water to a boil by placing it on a gas burner for a few minutes. Do this with the screw cap off. Then, when the water is boiling vigorously, suddenly screw the cap on, and then quickly place the can under a stream of cold water. The can will crumple up like so much paper. This spectacular experiment is one, which anybody can make at home but it is extremely convincing in demonstrating the production of a vacuum by the condensation of steam. Now remember, we wanted to create a vacuum at the exhaust end of the turbine. Now that we know how to create a vacuum, just how can we apply the principle to our turbine? Well, suppose we attach a large hollow vessel to the exhaust opening of the turbine and install a bank of small tubes in the vessel through which we can pump cold water. In this fashion – With such an arrangement, the steam issuing from the turbine will come in contact with the cold tubes and thereby turn back into water. This will create a vacuum in the vessel just as it did in the case of the gallon can. The vessel now, however, is made of heavy steel, capable of withstanding the pressure of the atmosphere, and will not collapse. Since cold water continues to flow through the tubes, the process is a continuous one. There will be a steady conversion of steam into water, and a steady state of vacuum will exist inside the vessel. Technically, such a vessel is called a condenser. Its purpose is twofold, first, to create a vacuum at the turbine exhaust, and second, to recover the condensate (the condensed steam) so that it can be used over again in the boiler. Since this condensate is really distilled water, it is very pure, and therefore, highly desirable for use as boiler feedwater. So, we build another smaller chamber at the bottom of the condenser to provide a place where the condensate can collect and from which it can be pumped back to the boiler, or rather, first to the boiler feedwater heater. This reservoir is called the hotwell, since the water, which collects in it, is fairly warm. Now, our system looks like this – Steam produced in the boiler flows through the main steam header to the turbine. In the turbine it passes, successively, through the various stages, losing pressure at each stage and giving up its energy to the blades on the rotor. This turns the electric generator and produces electricity. Emerging from the exhaust opening at the bottom of the turbine, the steam enters the condenser where it condenses on the tubes through which the cool circulating water flows. Condensation of the steam creates a vacuum which reduces the back pressure which otherwise would impede the flow of steam to a considerable extent. The condensed steam collects in the hotwell of the condenser and is drawn off by the condensate pump, which pumps the water into the feedwater heater. Here the water is further heated by the exhaust steam from the boiler feed pump (or other steam driven auxiliaries) and then is pumped back into the boiler by the boiler feed pump. The latter, it will be noted, is run by steam from the main high pressure header. We now have a completely closed system. All the water that is turned into steam in the boiler is condensed back into water in the condenser and pumped back into the boiler again. Of course, there are slight losses at various points in the system, leakage through pump bearings, steam leakage through valve packing, etc. To make up for this loss a small quantity of raw water has to be pumped into the system. This is known as make-up water or simply make-up. The idea is to keep the amount of make-up as low as possible. If you are interested only in the basic principle of a power plant, this is all you need to know. True, many more refinements can be added which will further improve the efficiency but the system shown in the last diagram would work, and its efficiency would not be too low depending upon the value of the steam pressure used. If this explanation, so far, has made sense to you and if it has been of interest, maybe you would like to go a little further and learn something about actual plants as they are built today. The boiler plant shown in the diagrams is fired by a chain-grate stoker. Most people are reasonably familiar with a stoker because many household furnaces burning coal have stokers. Power plant boilers are also fired by oil or pulverized coal. Where pulverized coal is used, the coal is first passed through a pulverizer which grinds the coal to the consistency of flour and then, by means of a fan, the powdered coal is blown into the furnace where it burns very much as a gas flame. Here is a simple diagram: Most of the large coal-fired plants today are fired by pulverized coal. The reason for this is that the control of pulverized coal firing is much more flexible than with stoker firing. With stoker firing there is always a bed of coal on the grate, which contains a considerable amount of heat. Even if the coal supply were cut off completely, the coal on the grate would continue to burn for an appreciable length of time. With pulverized coal there is no such reservoir of heat and if the coal supply is cut off combustion ceases instantly. The same is true of oil or gas. The use of pulverized coal instead of stokers does not change the basic principle of operation as far as the power plant as a whole is concerned; it merely involves different type of equipment. Now, what about steam pressure? In one of the foregoing paragraphs we said something about efficiency being related to steam pressure. Is this true? Yes, but largely because steam pressure is related to temperature. The higher the pressure of steam, the higher its temperature. At atmospheric pressure, that is, 15 lb. per sq. in. Absolute pressure, steam has a temperature of 212°F. At 500 lb. per sq. in. Absolute, the temperature of steam is 449°F. The efficiency of a turbine, or any other kind of heat engine such as a steam engine or a gas engine, does not depend upon the nature of the working medium – steam, compressed air, ammonia, etc. – but upon the quantity and the absolute temperature of the heat received and the heat rejected. This means merely that a turbine supplied with steam at 400 degrees and exhausting it at 212 degrees is more efficient than one receiving the steam at 300 degrees and exhausting at 212 degrees. Also, a turbine receiving steam at 400 degrees and exhausting at 212 degrees is less efficient than one receiving the steam at the same temperature (400) but exhausting it at 100 degrees. This not only explains the value of the condenser but also that of high steam pressure. Without the condenser, the lowest temperature at which steam can be exhausted is 212, since that is the temperature of steam at atmospheric pressure. By means of the condenser, however, a vacuum can be created so that the steam will exhaust at a pressure of, say, 10 pounds below atmospheric pressure, that is, at 5 lb. per sq. in. At 5 lb. per sq. in. Absolute, the steam temperature would be 162°F. The thing that is important in the operation of a turbine or any other kind of heat engine, then, is the temperature range through which the heat energy falls in its passage through the engine. The thermal efficiency of the engine depends upon this temperature range. This can be explained very simply by imagining a perfect engine – one in which there are no heat or friction losses of any kind. Of course, such a machine could never be built. Assuming that we had such an engine, however, let us connect it to a source of steam having a temperature of 400°F. Also, assume that the engine exhausts against atmospheric pressure. The exhaust steam then would have a temperature of 212°F. Now the thermal efficiency of such a perfect engine is easily figured by means of a very simple equation. Here it is: In this expression, E stands for efficiency in percent, T1, is the absolute temperature of the steam entering the engine, and T2 is the absolute temperature of the steam leaving the engine. So, with 400°F (860° Absolute)* for the entering steam and 212°F for the exhaust, the efficiency of this theoretical engine is: Instead of letting the steam from this engine exhaust against atmospheric pressure, suppose we attach a condenser to the exhaust opening so as to produce a back pressure of, say. 5 lb. per sq. in. As explained previously, at this pressure, the temperature of steam is 162°F. (162 + 460 = 622°Abs.) Now the equation will give us an efficiency of: So, by the addition of the condenser, we have raised the efficiency of the engine from 21.8 to 27.7 percent. *Temp. Absolute = Temp. F + 460. Absolute zero is 460° below zero F. This simple example, then, shows why power engineers have been striving, not only for higher and higher steam temperatures, but also for lower exhaust temperatures. The greater the range between the temperature of the steam entering and leaving the turbine, the higher will be the efficiency of the turbine. Of course, there is no such thing as a perfect heat engine and in practice the efficiencies obtained are much lower than those considered in this example, but the principle involved is essentially the same. It should be perfectly clear also that when a condenser is added to a system, it becomes necessary to pump large quantities of cooling water through it, and also, the condensate has to be pumped out of the condenser. This requires power and this added power has to be subtracted from that developed by the turbine when the efficiency of the system as a whole is considered. We do not get anything for nothing in this world. And this is as it should be; it makes the power engineers' job interesting regardless of which branch of the field he may specialize in. Take this quest for the attainment of higher steam temperatures and lower exhaust temperatures for example. This has lead the power engineer into all sorts of complex things involving chemistry, physics, metallurgy, techniques of manufacture and construction, and, of course as always, economics. High steam temperatures and pressures together with systems of higher and higher capacity make necessary alloy steels capable of withstanding the high temperatures and pressures; high rates of evaporation in boilers together with high pressures and temperatures make necessary elaborate feedwater treating systems involving constant, close chemical control; high pressures and temperatures also affect the character of the piping arrangements, valves, fittings, methods of insulation as well as many other things. With the use of high pressures and temperatures the simple method of heating the feedwater by exhaust steam from the boiler feed pump or other steam driven auxiliaries shown in the last version of our system will no longer suffice because the feedwater must be heated to a far higher temperature before it enters the boilers. Modern boilers operating at steam pressures of 1000 lb. per sq. in. have water temperatures of around 556°F. It is obvious that the feedwater will have to be somewhere near that temperature if severe strains are to be avoided. So we have the problem of increasing the feedwater temperature. Just how are we going to do it? What is the best way of doing it? Of course, one way of doing it would be to use high-pressure. High-temperature live steam from the main header and feed it into a special feedwater heater capable of withstanding the high pressure. This, however, would be wasteful. The steam in the main header is the most valuable commodity we have in the system, and every pound of it should be delivered to the main turbine where it can do the most good. Another method would be to supply the heat required for feedwater heating from a separately fired water heater but this would be even less efficient than taking live steam from the boiler itself. Suppose, however, that feedwater is heated by steam extracted from an intermediate stage of the main turbine; then power will be developed by the steam used for feedwater heating. Moreover, this additional power generated by the extracted steam will be produced at a very high efficiency and this, in turn, will increase the average efficiency at which the total power output of the system is generated. Now, why is this so? Why does this extracted steam produce power at a much lower fuel cost than that of power produced by steam flowing to the condenser? The answer to this question lies deep in thermodynamic theory but for practical purposes it can be explained as follows. In even the best modern condensing turbine power plants, approximately two-thirds of the heat present in the steam at the turbine inlet is left in the steam at the exhaust. Even if it were possible to have a 100 percent efficient turbine, the amount of heat thrown away at the exhaust would not be greatly reduced. Actually, this heat is carried away by the cooling water flowing through the condenser. This means, then, that even at best, only about one-third of the heat in the fuel can be turned into power. If however, we extract a portion of the steam from the turbine before it reaches the condenser, and use it to heat the feedwater, none of the heat in this steam will be wasted because it will all be absorbed in the boiler feedwater. Thus it decreases, heat unit for heat unit, the heat that must be supplied to the boiler. Putting it another way; of the steam flowing from the throttle to the condenser, about two-thirds of the heat will be thrown away, while of the steam flowing from the throttle to an extraction opening, no heat will be wasted. It follows directly, then, that the more power that can be generated by extracted steam, the higher will be the average plant efficiency. Of course, the amount of that which can be used for feedwater heating is determined by the amount of heat needed to raise the temperature of the boiler feedwater to the required level. This places an upper limit on the gain in efficiency, which can be made in this way. As in the previous diagrams, steam from the boiler enters the turbine and flows through the turbine stages, generating power. Most of the steam passes through the entire turbine and exhausts into the condenser. A part of the steam, however, is extracted from an intermediate stage of the turbine at a pressure and temperature higher than that at the exhaust. Here is the way in which we can take advantage of this scheme: In order that the temperature of the feedwater be high enough, it is necessary in this case to extract the steam at a point in the turbine where the temperature is somewhere in the neighborhood of the temperature of the water in the boiler. This, of course, limits the amount of power that can be obtained from this extracted steam. It would be better if the extraction point could be located closer to the exhaust end of the turbine. In heating water or any other substance to a higher temperature, however, only a portion of the total heat necessary need be at the highest temperature; much of the total heat is used in heating the water through a lower range of temperatures. With this fact in mind, it becomes possible to heat the feedwater successively in two or more stages in the manner shown here, Now, we have two feedwater heaters, a low pressure, low temperature heater receiving extracted steam from a stage close to the condenser, and another one receiving steam from an extraction point closer to the throttle. Water from the hotwell is first pumped to heater H1 where it is raised to a relatively low temperature and then it is pumped to the second heater where it is raised to a temperature close to the temperature of the water in the boiler. With this arrangement, it will be obvious; more power can be obtained from the total amount of extracted steam. In other words, by the use of two heaters instead of one, we have increased the efficiency of the system. So the question arises, if two heaters are better than one, why would not three be better than two, or four better than three? The answer, of course, is yes; each additional stage of extraction improves the thermal efficiency. However, in this instance, as in all engineering projects, there is a point of diminishing returns beyond which the further addition of heaters becomes uneconomical. Theoretically maximum efficiency would be obtained by means of an infinite number of extraction points and feedwater heaters. Actually, four or five stages are the economical limit and so most modern power systems are designed for this number. A few of the most modern stations use seven or eight. The method of heating the boiler feedwater in this fashion is known as regenerative feed-water heating; it is used in all modern steam power generating systems. With such systems a total of 20 to 30 percent of the throttle steam may be withdrawn from the turbine at various points and used to heat the feedwater. Here in this diagram we have incorporated four stages of feedwater heating in our system. At each heater the water is raised to a higher temperature. Since the pressure in each heater is higher than in the one preceding, a pump is necessary between successive heaters. Finally, after the water passes through the last heater, the boiler feed pump delivers it to the economizer section of the boiler. It is very evident from this diagram that a steam power plant can become quite complex when we try to take advantage of all the methods available to increase its efficiency. Indeed, the diagram, as shown, is still far from complete. While an air heater is shown, the fans and connection to the air heater pave been left out so as to keep the diagram simple. Actually, of course, the air is forced through the air heater by means of a fan, and after being heated it is forced into the furnace, in this manner: This shows how the forced draft fan forces cold air through the air heater and how the warm air is forced through ducts (which have to be insulated) into a plenum chamber underneath the stoker. Here it passes through the coal bed and thus supports combustion. A portion of the warm air is also admitted above the fuel bed. The air heater, it will be noted, is placed directly above the outlet of the furnace. Since it is built of a great many tubes it introduces a certain amount of resistance to the flow of the hot gases of combustion. In older plants where air heaters were not used a tall stack or chimney usually produced sufficient draft to pull the gases out of the furnace but where air heaters are used it has become universal practice to place an exhaust fan in the passage to the stack. Such fans are known as induced draft fans. Since these fans have to handle all the gases of combustion they often are very large and require powerful driving motors; sometimes rated as high as 1800 hp. The plant as we have, now designed it is a fairly good power plant but it lacks several important elements. One of these is a superheater. The purpose of a superheater is to heat steam above the temperature at which it is produced in the boiler. In practice, the superheater is merely an arrangement of alloy steel tubes placed in the gas path through the boiler. After the steam collects in the boiler drum it passes through the superheater tubes and is thus heated to a temperature higher than that associated with the pressure at which it is produced. For example, saturated steam at 1000 lb. per sq. in. absolute pressure has a temperature or 556°F. (Remember steam at atmospheric pressure has a temperature of 212°F.) Now by passing the 1000 lb. per sq. in. steam through the superheater, it can easily be heated to a temperature of 200 deg. higher, or 756°F. The pressure will remain the same. Such superheated steam has two advantages over steam that is not superheated; first, it increases the thermal range of the steam cycle, and hence the efficiency, and second; being dryer, it is less likely to condense in the lower stages of the turbine. In large turbines, the formation of drops of water on the blades near the exhaust end of the turbine can be quite damaging. By the use of superheated steam, however, this condensation can be minimized to a point where it is harmless. Hence, all modern power plants use superheated steam, indeed in the most modern plants in addition to the superheater the steam is reheated in a second superheater after it has passed through a portion of the turbine. With such an arrangement steam collected in the steam drum of the boiler passes first through the primary superheater, then through the first few stages of the turbine, then back to the boiler where it is reheated in the reheat superheater, and finally, sent back to the lower pressure stages of the turbine and so, to the condenser. The idea is outlined here: As a rule, the reheated steam has a temperature only a little below that of the primary steam but the pressure is considerably lower. As an example, the primary steam may have a pressure of 1400 lb. per sq. in, and a temperature of, say, 1050°F. After this has passed through a number of stages in the turbine it is extracted at, say, 200 lb. per sq. in. where its temperature will be around 400°F. In the reheat section of the boiler this steam will then be heated to a temperature of, say, 1000°F. Its pressure, however, will still be 200 lb. per sq. in. So now we have superheat, reheat, and regenerative feedwater heating in our system and it is becoming very efficient, as steam cycles go. With systems of this kind thermal efficiencies of around 32 percent can be obtained, that is, some 32 percent of the heat energy in the fuel will be converted into electrical energy. You may wonder why this figure is so low. The reason, as implied earlier in this treatise, lies in the fact that most of the heat is carried away by the condenser cooling water. Unfortunately, this low efficiency is a consequence of the second law of thermodynamics and there is practically nothing that engineers can do about it. If the temperature of the steam at the exhaust of the turbine could be brought down to absolute zero, we could recover most all of the heat energy in the fuel but since we live in a world where the ambient temperature is some 490°F. above absolute zero there is no way by which we can ever reduce the temperature of the exit steam below ambient temperature. Of course, we could do it by refrigeration but the refrigeration system would require power and so we would gain nothing, indeed we would lose efficiency. We have now incorporated in our system about all the known methods for improving the efficiency. Further improvement can be made in any system by going to still higher pressures and temperatures, by refinements in the condensing system, and by the further reduction of heat losses wherever they occur by improved equipment, insulation, and recovery of minor wastes, but the gains to be expected by these means are relatively small. In large power stations, however, each increment, each fraction of a percent in the overall efficiency is worthwhile because such stations use hundreds of thousands, indeed, millions of tons of coal a year, and even small gains in thermal efficiency reflect large savings in fuel costs. As indicated at the beginning of this discussion, today, modern steam-electric generating stations use less than one third the amount of coal for the same kilowatt output that they did 30 years ago. This saving has been brought about by continual refinement such as described in these pages. It is obvious then, that the operation of a power station, every effort has to be made at all times to keep the temperatures, pressures, and the vacuum in the condenser at their optimum values. A change in anyone of these values affects the efficiency of the system as a whole. In the condenser, for example, the vacuum must be maintained at its highest value by the use of the coldest possible circulating water. Since the circulating water usually is drawn from rivers or lakes, the temperature varies throughout the year, warming up in summer and becoming cooler in winter. Since the condenser is under vacuum there, is a tendency for slight air leakage into the system and unless this air is removed it tends to accumulate in the condenser and thus impair the vacuum. So condenser air pumps are provided which continuously remove the air. These pumps separate the air from the water vapor and return the water to the condensate system. These condenser air pumps, nowadays, are of the steam ejector type. Even these are fitted with heat exchangers to recover the heat that might otherwise be lost in extracting the air from the condenser. One element in power station operation that has received an ever-increasing amount of attention in recent years is that having to do with the delivery of pure feedwater to the boiler. Modern high pressure boilers evaporate a million or more pounds of water per hour, and they do this 24 hours a day, 365 days a year. With such tremendous rates of evaporation, it is obvious that if the water delivered to such boilers contained even small amounts of scale-forming materials, the internal heating surfaces of the boilers would soon become so coated with scale that overheating and subsequent failure would result. Because of this, elaborate systems of chemical feedwater treatment have been developed which not only reduce the scale forming materials to practically zero but also reduce the oxygen content to reduce corrosion. Oxygen in hot water is an extremely corrosive agent. Sometimes these feedwater treatment systems involve evaporators in which raw water is evaporated by steam extracted from the turbine and then condensed. Other systems use ion exchange type demineralizers, or chemical treatment, which precipitate the scale forming materials in the form of sludge before it enters the boiler. Whatever the type of system, the purpose is to add only absolutely pure water to the system. It should be obvious that the amount of water that need be added to the system while it is in operation is small, since all the steam flowing through the turbine is condensed and returned to the boiler. There are small losses, however, these have to be replaced by what is known as make-up water. This make-up is the water that has to be treated. Another factor in the operation of large power stations that has become important is the emission of dust and fly ash from the stacks. In plants where many thousands of tons of coal are burned a day, the emission of fine ash from the stacks can become a serious nuisance and in many communities ordinances have been enacted requiring power stations to reduce the emission of fly ash and dust to a minimum. This has not been an easy problem and many hundreds of thousands of dollars have been spent in developing equipment for collecting such dust before it enters the stacks. A number of different methods are in use. In one type of system, the flue gases are passed between electrically charged plates. The intense electrostatic field charges the dust particles causing them to be attracted to the plates. After the dust has accumulated to a certain thickness on the plates it is scraped or knocked off. In other systems, the flue gas is whirled through cyclone separators or passed through sprays of water. In any case, whatever system is used, they are expensive and involve high orders of engineering skill in their design. So far, we have been concerned only with the steam end of the power system; indeed, we have only mentioned the electric generator in passing. This is due to the fact that most of the economies that can be made in the operation of a steam-electric generating station lie on the steam side. The modern electric generator is an extremely reliable and efficient machine. Modern generators have efficiencies of as high as 97 percent so the additional gains that can be made in the improvement of generators is small. As pointed out at the beginning of this treatise, basically an electric generator consists of a magnet spinning inside a group of coils of wire. As the rotating magnetic field cuts the convolutions of the stationary coils, electric currents are set up in the coils, and by properly connecting them currents of almost any required voltage can be produced. Modern generators produce current anywhere between 13,000 and 22,000 volts. Where the electricity has to be transmitted over long distances – over 15 or 20 miles, – the generator voltage is stepped up by means of transformers. A transformer is a sort of electrical lever by means of which voltages can be stepped up or reduced to any desired value. Transformers are the most efficient large machines man has yet devised, some of the larger units having efficiencies as high as 99 percent. Now, in the light of all this let us finally redraw the diagram of our power plant to incorporate everything we have mentioned. As you see, it has become a highly complex affair involving many branches of science – physics, chemistry, metallurgy, thermodynamics, hydraulics, structural engineering, electricity, etc. The drawing has become so large, that it has been necessary to put it on a separate double size sheet. Complex as it may seem, this diagram is still only the simplest of schematic diagrams; an actual power plant layout has in it count less other small and large devices and sub-systems not shown or only indicated on this drawing. For example, so far in this treatise nothing has been said about the combustion control system. This is indicated on this diagram merely in bare detail. Showing only the basic devices. Actually the combustion control system in a large plant would require a large separate drawing since it is very important and the operation of the station would be virtually impossible without it. Essentially it consists of a device, which is sensitive to slight variations in steam flow. This device, in turn, controls a variety of relays and actuating mechanisms, which automatically control the flow of fuel, air, and water to the boiler in accordance with the varying load conditions. The primary connection of the combustion control system to the main steam line is shown at A. The diagram also incorporates reheat and both high and low pressure turbines. After the steam has passed through the high-pressure turbine, it is returned to the boiler and, after being heated again, it is delivered to the low-pressure turbine. Also, there are two sets of water heaters, three low-pressure heaters taking extraction steam from the low-pressure turbine, and two high-pressure heaters using extraction steam from the high-pressure turbine. Between, the two sets of heaters is a de-aerating heater in which the oxygen in the feedwater is boiled off. Oxygen in boiler water at high pressures is extremely corrosive and must be removed before it is delivered to the boiler. This is done by means of a de- aerating heater, which, in effect, is merely a large tank of water boiling under atmospheric pressure. The necessity for proper feedwater treatment has already been mentioned. On the diagram, the feedwater treating system is merely indicated. It consists of a chemical treating system feeding into the plant system through an evaporator. Thus, all the make-up into the plant system passes through the evaporator. This make-up water (the water which has to be added to the system continually to replenish water lost by leakage and blowdown) enters the system in the form of vapor that is delivered, as shown, to the de-aerating feedwater heater. In the evaporator the incoming water is heated by exhaust steam from the boiler feed pump turbine or other steam driven auxiliaries. As already mentioned, a demineralizer may be used instead of an evaporator. Note that the boiler feed pump turbine receives steam from the main steam header through a reducing valve and a desuperheater. It is not usual to use superheated steam for small auxiliary turbines, for several reasons, first because the metals needed with super-heated steam are very expensive, and second, high thermal efficiency is not so important in an auxiliary turbine since the heat in the exhaust is returned to the system. It becomes expedient, therefore first to reduce the high-pressure steam from the main steam system to a lower pressure by means of a reducing valve and then to desuperheat the steam by spraying water into it. In this way, low-pressure, saturated steam is delivered to the auxiliary turbines. The diagram, it may be noted, shows a symbol labeled "continuous blowdown". Because of the continual recycling of the water through the boiler and because of slight leakages in the system, the boiler water tends to increase its concentration of impurities – scale-forming salts. To keep this concentration to a minimum, it is necessary to blow down the boiler periodically or continuously. In small power plants, the operator does this periodically by merely opening a blow-off valve for a few seconds and blowing out the water in the lowest part or the boiler where the concentration is highest. In large plants, the amount of heat lost by such blowdown practice tends to be rather high, so continuous blowdown systems are used. With such systems a small amount of water is withdrawn continuously but before it is run to the sewer, it is run through a heat exchanger in which the heat from the blowdown is transferred to the incoming feedwater. These are some of the thousand and one details of a modern power plant that make it the complex thing it is. It is not the intent here to consider all these details but merely to point out that they exist. Little has been said about control except brief mention concerning the combustion control. The latter, however, constitutes only one element of the station control as a whole. Today, most power stations are controlled from a single control room where quantities from all parts of the plant are measured, indicated, recorded and integrated. The modern generating station is one of the most completely automatic systems man has devised; it has to be because it would be virtually impossible for operators to watch and accurately control all the varying quantities involved in the operation of a plant. Indeed, it has become impossible to keep track of all the pressures, temperatures, liquid levels, speeds of all the various machines and recently a completely automatic supervisory system for recording and announcing all the hundreds of items involved has been developed. This system, called SARA from its full name, Sequential Automatic Recording Annunciator, is an electrical system using techniques similar to those used in electric computer practice. The protective features of the station are complex and involved. In case of trouble due to failure of a piece of apparatus or an electrical fault on the external electrical system events happen rapidly. If the load on a large generator suddenly dropped and the turbine governors failed to act, the machine, normally rotating at 3600 rpm, would suddenly increase its speed and would explode from centrifugal force in a matter of three seconds. The 40-ton rotor of a modern electric generator spinning at 3600 rpm has a rotational energy of 650 million foot-pounds. This is approximately the same kinetic energy that a 40-ton jet airliner would have at a speed of 500 miles per hour. On the boiler side, if the feedwater supply failed, the boiler, producing, say, a million pounds of steam per hour would run dry in 90 seconds. Since these equipments are valued in terms of millions of dollars, it is obvious that every possible measure must be taken to insure their protection. There are complex instruments that not only measure the speed of a turbogenerator to a fraction of a revolution, but also to measure the degree of shaft eccentricity, and the vibration characteristics of the machine. An expansion indicator shows the axial expansion of the turbine casing. The diagram, it will be noted, shows the generator somewhat differently than in the early diagrams – here it is shown symbolically. Also, it is associated with an exciter, a device that, so far, has not been mentioned. Its purpose is to supply the magnetizing current for the rotating magnet. In an actual electric generator the rotating magnet is not simply a permanent magnet such as is indicated in the earlier diagrams. A permanent magnet would not provide a strong enough magnetic field; so an electromagnet is used. An electromagnet is magnetized by electricity flowing through coils wound around the magnet structure. In the case of a large generator a very powerful direct current is sent through the coils on the rotor. These windings are known as the field coils, since they produce the magnetic field in the generator. To supply the field coils with current makes it necessary to provide an arrangement of sliding contacts to conduct the current from the stationary to the rotating part of the machine. Also, it becomes necessary to provide a separate source of current to excite the field coils. Basically, here is how it is done, though in an actual generator the shape of the rotor is quite different from the simple bar magnet shown in this diagram. Two collector rings are mounted on the main generator shaft as shown. These rings are connected to the ends of the field winding. Stationary brushes mounted on the collector rings, thus conduct the exciting current from the source to the windings on the rotating member of the generator. The direct-current needed to excite the field winding is usually supplied by a separate small direct- current generator mounted on the same shaft as the main rotor. Such a small d-c generator is called the exciter because it furnishes the excitation current for the main generator. The exciter circuit also provides a means of controlling the voltage of the main generator. By varying the excitation current by means of the field rheostat, the voltage of the generator can be controlled between normal operating limits. The speed of the generator, however, is controlled by the steam-turbine governor and this control is exceedingly close. Indeed the generator speed is so closely controlled that it serves as our time standard. Most of us these days measure our time by means of synchronous electric clocks. These clocks depend upon the constant speed of the generator. Means are provided to check this speed in relation to accurate independent clocks at the National Bureau of Standards. Temperature recorders measure the temperature deep in the interiors of the windings of the generator, differential relays guard against internal electrical failure of the machine. Elaborate hydrogen cooling systems are provided to remove the heat losses from the generator and by means of hydrogen coolers in the condensate system, this heat is returned to the feedwater. No heat is lost that possibly can be saved. Even the heat received in the bearings is returned to the feedwater by means of the oil coolers in the condensate system. From all this, it should be clearly evident that a modern power plant engineer has to be a specialist of a very high order. While the system operates automatically it still needs the guidance of engineers who are thoroughly conversant not only with details of the equipment but also with the basic principles upon which it operates. Power engineering is a field calling for the highest engineering talent in both design and operation. It provides an occupation that is at once stimulating and challenging, and at the same time, affords steady employment in a rapidly growing field with practically unlimited opportunity. Also, despite this rather long treatise on power plant design and operation, it should be obvious that it is, basically, still only a primer. |
| Surplus Generation Equipment, LLC, Brokering the following surplus equipment: Natural gas generator, nat gas generator, micon wind turbine, Gas Turbine, Frame 3 Turbine, Frame 5 Turbine, Frame 7 Turbine, Frame 9 Turbine, 7ea Turbine, 7fa Turbine, LM2500 Turbine, LM5000 Turbine, LM6000 Turbine, Siemens 501 Turbine, GE Turbine, Alstom Turbine, Solar Turbine, Rolls Royce Turbine, Mitsubishi Turbine, Westinghouse Turbine, Solar Taurus Turbine, Solar Mars Turbine, Solar Centaur Turbine, Solar Tornado Turbine, Steam Turbine, Gas Turbine, Used Steam Turbine, Surplus Steam Turbine, Used Gas Turbine, Surplus Gas Turbine, Simple Cycle Turbine, 10MW Turbine Generator, 20MW Turbine Generator, 30MW Turbine Generator, 40MW Turbine Generator, 50MW Turbine Generator, 60MW Turbine Generator, 70MW Turbine Generator, 80MW Turbine Generator, Warstsila, Caterpillar, Diesel Generator, Diesel Genset, Natural Gas Generator, Natgas Generator, Natgas genset, Jenbacher, Jenbacker, Cooper, Cooper Superior, Compressor, Cooling Tower, Wind Turbine, Micon, Micon Wind Turbine, Vestas, Vestas Wind Turbine, Deutz, Deutz generator, Used Wind Turbine, Used Diesel Generator, Wartsila |

| Surplus Generation Equipment |

Search Site For
Manufacturer
Manufacturer
Here are some additional resources to aid you in your search for
generation equipment feel free to contact us if you have any questions.
generation equipment feel free to contact us if you have any questions.
Providing Surplus & Used
Power Generation Equipment
Worldwide - 770.715.5442
Power Generation Equipment
Worldwide - 770.715.5442